Neocate LCP, the UK's No.1 AAF1
Neocate LCP is a hypoallergenic amino acid formula (AAF) designed to meet the nutritional needs of infants who have Cow’s Milk Allergy (CMA), Multiple Food Protein Allergies (MFPA) or require an AAF from birth.

Fast and effective Cow's Milk Allergy symptom resolution in 3-142,3 days

Within 3 days on Neocate LCP, studies showed significant improvement of eczema and disappearance of other symptoms (e.g. vomiting, diarrhoea)2.
Within 14 days on Neocate LCP, studies showed resolution of all gastrointestinal symptoms. 100% of infants gained weight whilst receiving the AAF3.

Over 35 years of studies and research support Neocate LCP
Neocate LCP is the only AAF with over 35 years clinical experience and is the most researched formula in the UK market1.
More publications (>95) support the use of Neocate LCP than any other hypoallergenic amino acid-based product1.

Neocate LCP can resolve symptoms that persist on an extensively hydrolysed formula (EHF)4
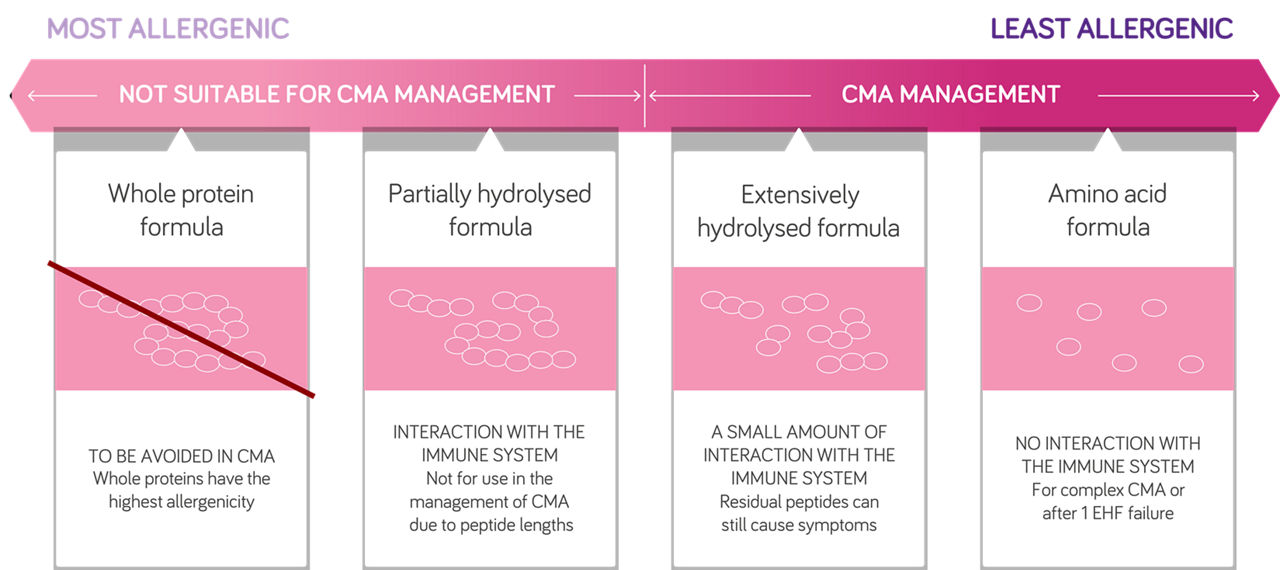
Up to 10% of infants with uncomplicated CMA and up to 40% of infants with more complex allergy may not tolerate an EHF.4 Guidelines suggest that symptoms should resolve within 2-4 weeks on an EHF in most infants, however, where symptoms persist on an EHF, or in formula fed infants with severe CMA, an AAF should be considered.5
Neocate LCP nutritional information
- nutritionally complete9
- hypoallergenic11
- supports growth6, 10, 11
- suitable as a sole source of nutrition up to the age of 1 year
- contains long chain polyunsaturated fatty acids (LCPs) in line with EFSA recommendations for the visual development of infants and in a 1:1 ratio7,8
- Halal certified
- Kosher approved
- can be used in combination with breast milk and/or solids during or after weaning
- is part of an age-adapted range of products
For a detailed nutritional breakdown, please view the data card.
What is the difference between Neocate LCP and Neocate Syneo?
Neocate LCP does not contain the live beneficial bacteria Bifidobacterium breve M-16V. This means Neocate LCP can be used in patients where an AAF containing live bacteria would be contraindicated.
Neocate Syneo is the only† amino acid-based formula to contain a synbiotic blend of both prebiotics and probiotics*.
† Product can be provided to patients upon the request of a Healthcare Professional. They are intended for the purpose of professional evaluation only.
- Data on file, 2023.
- De Boissieu et al. Allergy to extensively hydrolysed cow milk proteins in infants: Identification and treatment with an amino acid-based formula. J Pediatr 1997;131(5):744-7.
- Vanderhoof, J.A. et al. Intolerance to protein hydrolysate infant formulas: An under recognized cause of gastrointestinal symptoms in infants. J Pediatr 1997;131(5):741-4.
- Hill DJ et al. The efficacy of amino acid-based formulas in relieving the symptoms of cow's milk allergy: a systematic review. Clin Exp Allergy 2007;37(6):808-22.
- Venter C et al. Diagnosis and management of non-IgE-mediated cow’s milk allergy in infancy - a UK primary care practical guide. Clin Transl Allergy 2013;3(1):23.
- Isolauri E et al. Efficacy and safety of hydrolyzed cow milk and amino acid-derived formulas in infants with cow milk allergy. J Pediatr 1995;127(4):550-7.
- European Food Safety Authority (EFSA). DHA and ARA and visual development: Scientific Opinion of the Panel on Dietetic Products, Nutrition and Allergies. EFSA J 2009;941:1-14.
- Harbige LS. Fatty acids, the immune response, and autoimmunity: a question of n-6 essentiality and the balance between n-6 and n-3. Lipids 2003;38(4):323-41.
- Commission Directive 1999/21/EC (FSMP) and Commission Directive 2006/141/EC (as laid down in the infant formulae and follow-on formula guidelines).
- Burks AW et al. Synbiotics-supplemented amino acid-based formula supports adequate growth in cow's milk allergic infants. Pediatr Allergy Immunol 2015;26(4)316-22.
- Harvey BM et al. Effects on growth and tolerance and hypoallergenicity of an amino acid-based formula with synbiotics. Pediatr Res 2014;75(2):343-51.
Accurate at time of publication, October 2023.
Help us provide information most relevant to you
Please ensure your role and areas of interest are up to date.



