Aptamil Comfort's ingredients, clinical evidence, parent and Healthcare Professional feedback

Aptamil Comfort's tailored ingredients
Aptamil Comfort has a tailored combination of ingredients to support GI health and promote a healthy gut microbiota for the dietary management of colic and constipation
Tailored fat blend with high levels of beta-palmitate may help to promote softer stools and aid the absorption of fat and calcium1–3
Partially hydrolysed whey protein for easy digestion and reduced gastrointestinal transit time4,5
Prebiotic oligosaccharides – short chain Galacto-oligosaccharides and long chain Fructo-oligosaccharides (scGOS:lcFOS) (9:1) supports a healthy gut microbiota by increasing the number of beneficial bacteria and reducing harmful bacteria6,7
Reduced lactose can help to reduce flatulence and abdominal discomfort8
Large pool of clinical evidence
Aptamil Comfort benefits from a large pool of clinical evidence with proven efficacy for the management of colic, constipation and related symptoms.

| Reference | Study design & population | Groups | Study period | Outcomes (Comfort vs. control) |
| Veitl et al. 2000 | RCT with infants < 3 month with at least one of the following symptoms: excessive crying, constipation (with or without cramps), and general abdominal distress | Comfort (n=34) Control (n=17) | 2 weeks |
|
| Schmelzle et al. 2003 | RCT with healthy term infants < 2 weeks | Comfort (n=49) Control (n=53) | 10 weeks |
|
| Savino et al. 2003 | Prospective observational study with infants < 3 months infantile colic and/or constipation | Comfort (n=604) | 2 weeks |
|
| Savino et al. 2005 | RCT with infants < 4 months with infantile colic | Comfort (n=55) Control (n=40) | 2 weeks |
|
| Savino et al. 2006 | RCT with infants < 4 months with infantile colic | Comfort (n=103) Control (n=93) | 2 weeks |
|
| Bongers et al. 2007 | RCT (cross-over design) with infants 3-10 weeks with constipation | Comfort (n=38) Control (n=38) | 6 weeks |
|
Proven Efficacy
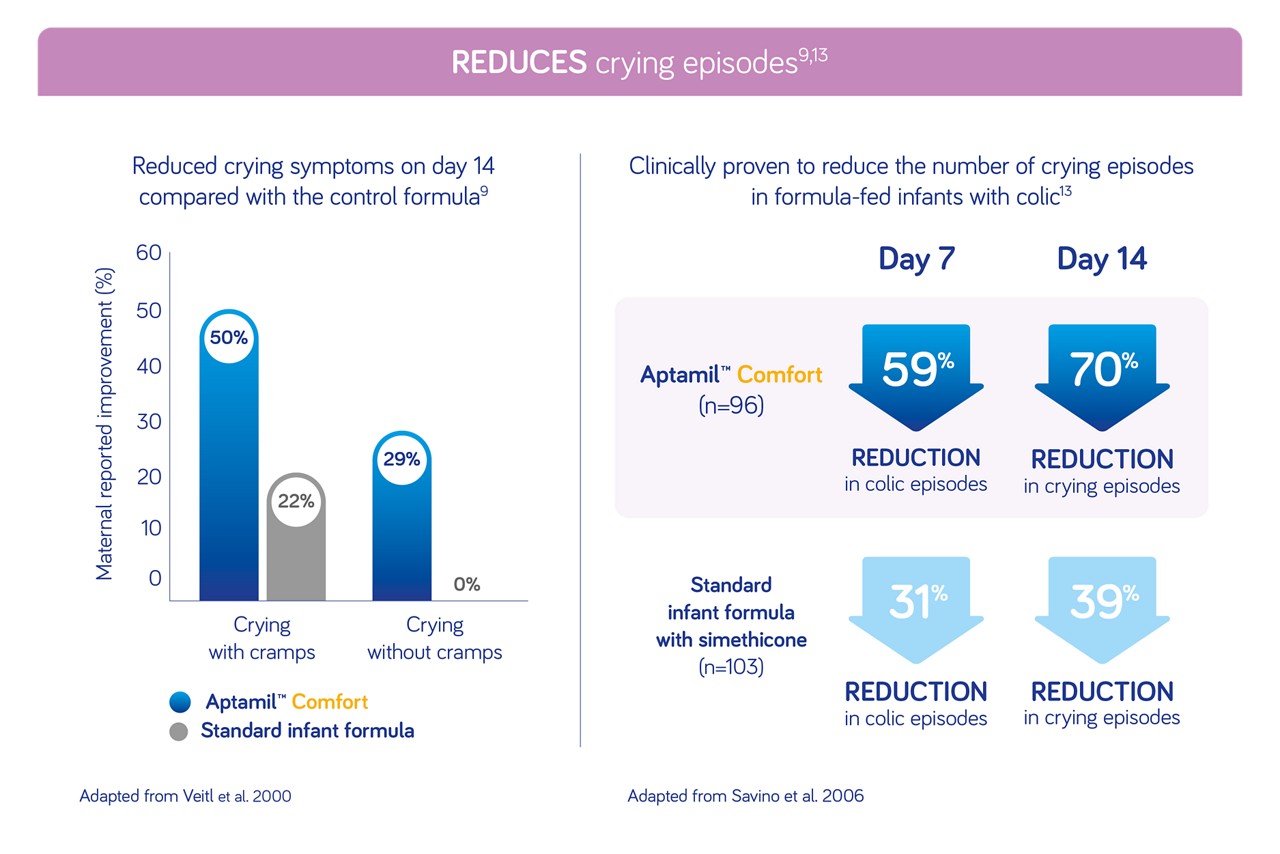
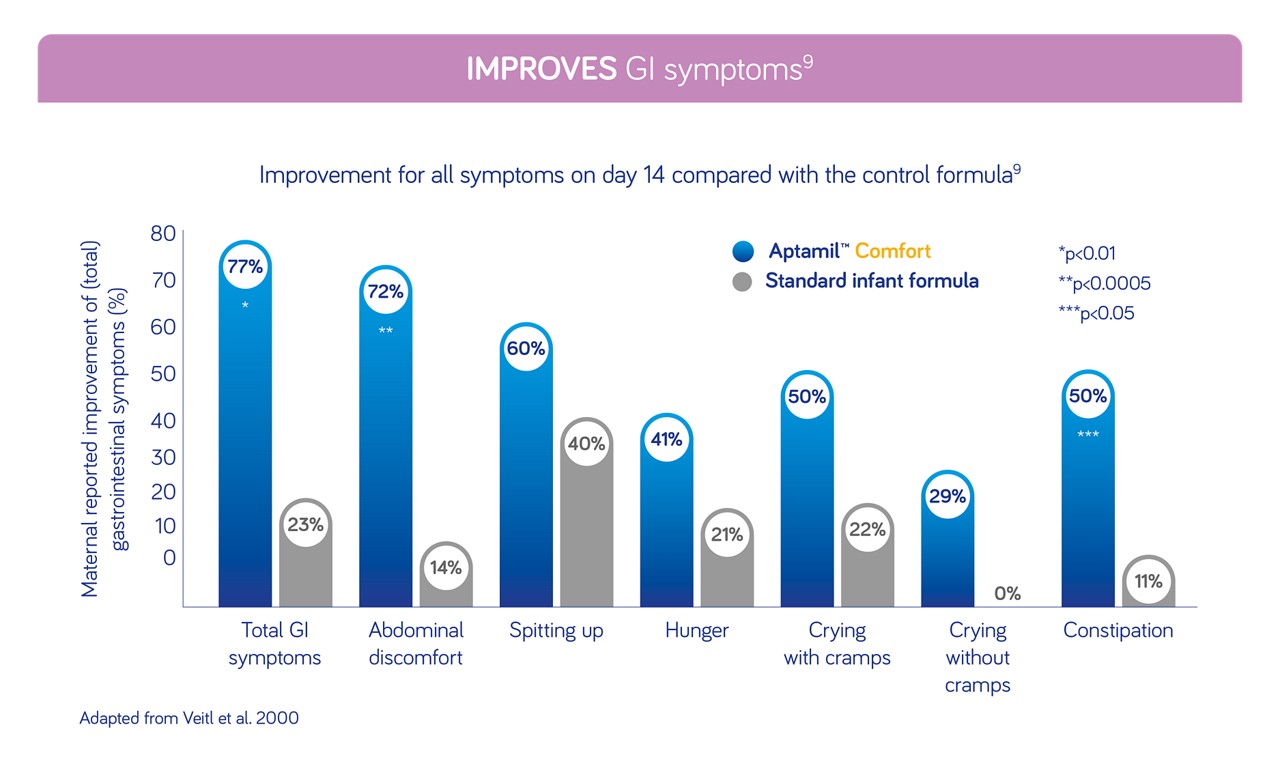
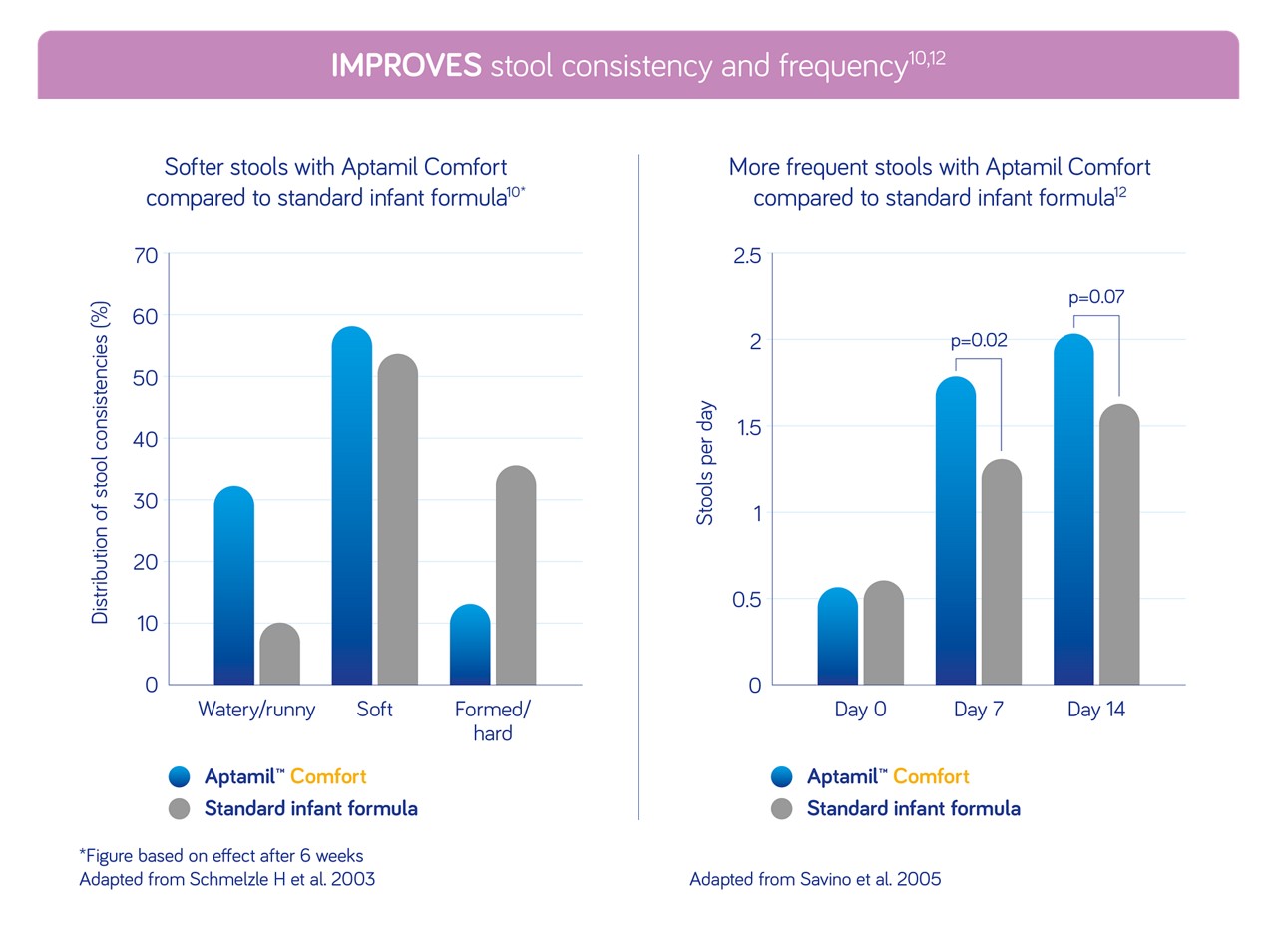
Rated by parents and healthcare professionals
Aptamil Comfort provides HCP & Parental satisfaction*
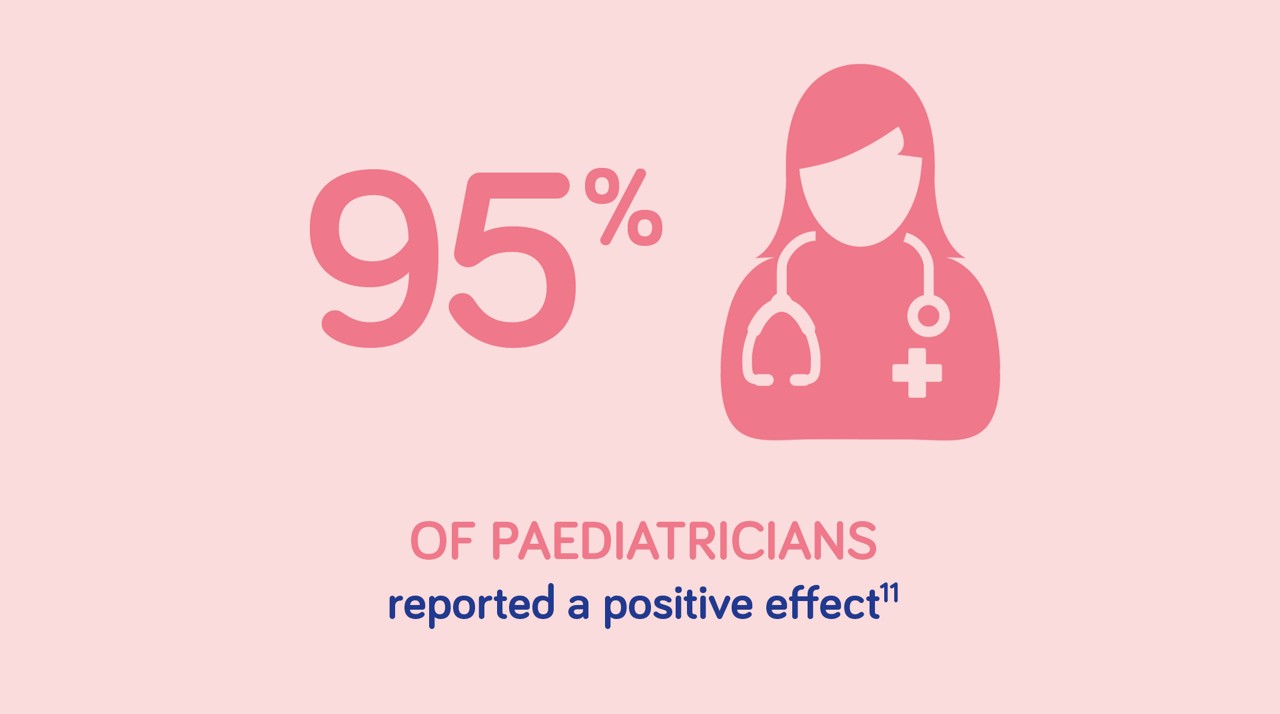

*based on an observational study conducted in Italy, with 604 infants. Adapted from Savino et al. 2003
- Havlicekova Z et al. Nutr J. 2016;15:28.
- Carnielli VP et al. Am J Clin Nutr. 1995;61:1037–1042.
- Kennedy K et al. The American Journal of Clinical Nutrition. 1999;70.5: 920–927.
- Billeaud C et al. Eur J Clin Nutr. 1990;44(8):577–583.
- Tolia V et al. J Pediatr Gastroenterol Nutr. 1992;15(3):297–301.
- Knol J et al. J Pediatr Gastroenterol Nutr. 2005;40(1):36–42.
- Moro G et al. J Pediatr Gastroenterol Nutr. 2002; 34(3):291–295.
- Kanabar D et al. J Hum Nutr Diet. 2001;14:359–363.
- Veitl et al. Ernährungsmed. 2000;2:14–20.
- Schmelzle H et al. J Pediatr Gastroenterol Nutr. 2003;36(3):343–351.
- Savino et al. Acta Paediatr Suppl. 2003;91:86–90.
- Savino et al. Acta Paediatr Suppl. 2005;94:120–124.
- Savino F et al. Eur J Clin Nutr. 2006;60:1304–1310.
- Bongers et al. Nutr J. 2007;6:8.
- Scholtens PA et al. World J Gastroenterol. 2014;20(37):13446–13452.
Help us provide information most relevant to you
Please ensure your role and areas of interest are up to date.
