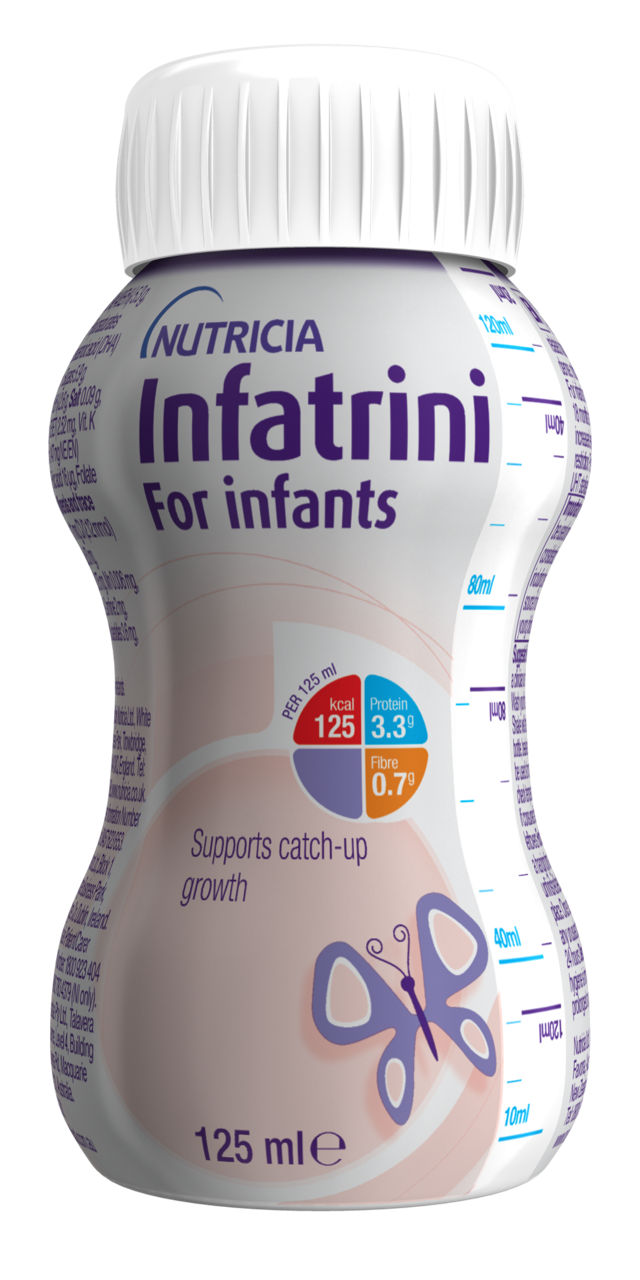
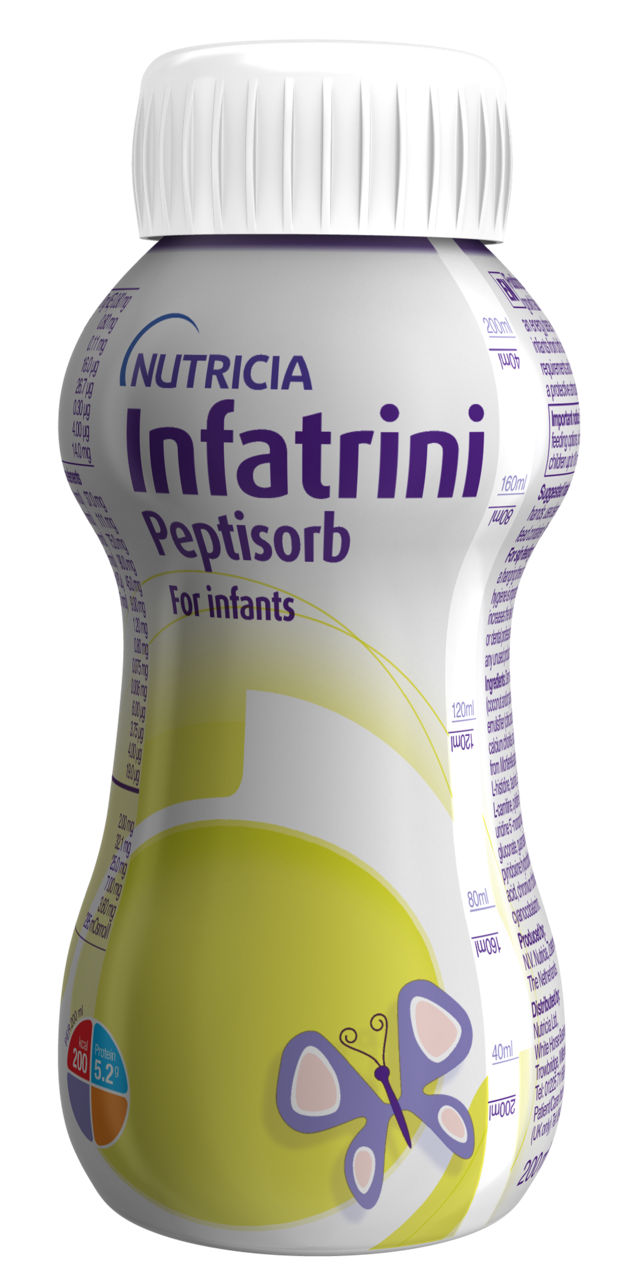
Infatrini Range
The Infatrini range are Foods for Special Medical Purposes for use under medical supervision. Infatrini products are nutritionally complete, energy dense 1kcal/ml, ready to use feed for the dietary management of infants (from birth up to 18 months or <9kg in body weight) with faltering growth, or who have increased nutritional requirements and/or require fluid restriction.

About Infatrini Range
The importance of early intervention
Nearly 1 in 2 paediatric patients with chronic diseases are malnourished1. Undernutrition in early life may not only result in impaired immune function, increased risk of infection, poor wound healing and GI dysfunction, but may lead to faltering growth and may delay normal feeding development and the onset of puberty2-10.
Poor weight gain from birth to 6-8 weeks of life is a stronger predictor of developmental delay than poor weight gain over the remainder of the first year, highlighting the importance of acting quickly11.
Consequences of not intervening early
Effect on cognitive development and brain function: Faltering growth and undernutrition in infancy have been associated with cognitive and behavioral impairment during childhood and adulthood. Furthermore, studies in term infants have shown that undernutrition during infancy is associated with permanent alterations in brain growth and function12-15.
Impact on IQ: Children with faltering growth achieve IQ scores of 4.2 points lower than children with a history of adequate growth16.
Physical impact: Muscle has a critical period of development before birth and during the first 6 months afterwards. Failure of growth at this time is associated with a life-long reduction in muscle17.
Additional product information

WHY CHOOSE A WHOLE PROTEIN FORMULA WHEN TOLERANCE ISN’T AN ISSUE?
- Where a high energy infant formula is indicated, a whole protein formula is closer to breast milk. The protein structure in human milk as well as weaning food is almost all whole protein18.
- Whole proteins support gut maturation. Evidence suggests a role for whole protein in stimulating the development and maturation of the gut19,20.
- Lack of evidence for use of partially hydrolysed formula in faltering growth with no tolerance issues. Whole protein formulas have been used in infants with faltering growth for decades and have been demonstrated to be well tolerated21,22. There is a lack of data to support the routine use of partially hydrolysed formula as an alternative option to human milk or whole protein, in infants with faltering growth who have no tolerance issues19-23.
WHY CHOOSE AN EXTENSIVELY HYDROLYSED FORMULA WHEN TOLERANCE IS AN ISSUE?
- Infants with complex medical conditions and malabsorption or maldigestion will often fail to tolerate a whole protein formula24.
- An extensively hydrolysed protein formula is indicated in complex conditions. In patients with complex medical conditions, extensively hydrolysed formulas are routinely used8 and promote good tolerance24,25 while partially hydrolysed formulas may not be any better tolerated than whole protein26.
- Extensively hydrolysed protein promotes faster gastric emptying. Extensively hydrolysed protein has been demonstrated to promote faster gastric emptying than partially hydrolysed or whole protein27.
Infatrini Range
- Diamanti A et al. Clin Nutr 2019:1877-82.
- Phornphatkul C et al. Clin Pediatr 1994;33:147-54.
- Jahari AB et al. Eur J Clin Nutr 2000;54:S60-8.
- Jahari AB et al. Eur J Clin Nutr 2000;54:S74-9.
- Berkman DS et al. Lancet 2002;359:564-71.
- Pozo J et al. Best Pract Res Clin Endocrinol Metab 2002;16:73-90.
- Reid M et al. Am J Clin Nutr 2002;76:1409-15.
- Li H et al. Am J Clin Nutr 2003;77: 498-505.
- Joosten KFM et al. Curr Opin Pediatr 2008;20:590–6.
- Sullivan PB Eur J Clin Nutr. 2010;64 Suppl 1:S1.
- Drewett R et al. J Reproduct Inf Psychol 2005;23:309-17.
- Chang SM et al. Dev Med Child Neurol 2010;52: 831-6.
- El-Sherif AM, Babrs GM, Ismail AM. Life Sci J 2012; 9: 738-742.
- Pongcharoen T et al.. Arch Pediatr Adolesc Med 2012;166: 411-6.
- Galler JR et al. J Nutr 2012b; 142: 788-94.
- Corbett SS, Drewett RF J Child Psychol Psychiatry 2004; 45 (3): 641-654
- Baker D.J.O Journal of Nutrition 2007;137:1058-1059
- Lönnerdal B. Nestle Nutr Inst Workshop Ser. 2016;86:97-107.
- Vandenplas, Y, et al. J. Pediatr. Gastroenterol. Nutr., 2016;62(1):22-35.
- Senterre T, Rigo J. Nestle Nutr Inst Workshop Ser. 2016;86:39-49.
- Clarke, S.E et al. J Hum Nutr Diet, 2007;20(4):329-339.
- Evans, S., et al. J Hum Nutr Diet, 2006;19(3):191-197.
- Fleischer DM, et al. Nestle Nutr Inst Workshop Ser. 2016;86:51-65
- Smith, C., et al. Clin Nutr, 2018;37(3):1005-1012.
- Marino, L.V., et al. J Hum Nutr Diet. 2019;32(3):400-408.
- Savage, K., et al. JPEN. 2012;36:118S-123S.
- Staelens, S., et al. Clin Nutr, 2008;27(2):264-268.
Looking to speak to your local Nutricia representative?
Select ‘request a meeting/ call with a local sales rep’ as the topic of enquiry when completing our contact form, and your local account manager will be in touch shortly.